Tag: 3dfm
3DFM NanoRod Bead Vortex
This clip starts with a bead being positioned near a nanorod using a laser trap. Then, an orbit is applied to the rod using the magnets of the 3DFM and it starts to beat. A flow in the CCW direction is created, and the bead can be seen to flow around the rod. Then, the beat frequency is doubled, and the bead’s orbital radius increases.
Perfect Orbit
We observe a magnetic bead with a 4.4-micron diameter being pulled around by the four poles of the initial “”Betty”” setup of our 3DFM. The bead is in a corn-syrup solution with a calibrated viscosity of 86 centiPoids. The video shows two complete orbits of the bead, as it is pulled towards each magnet in turn.
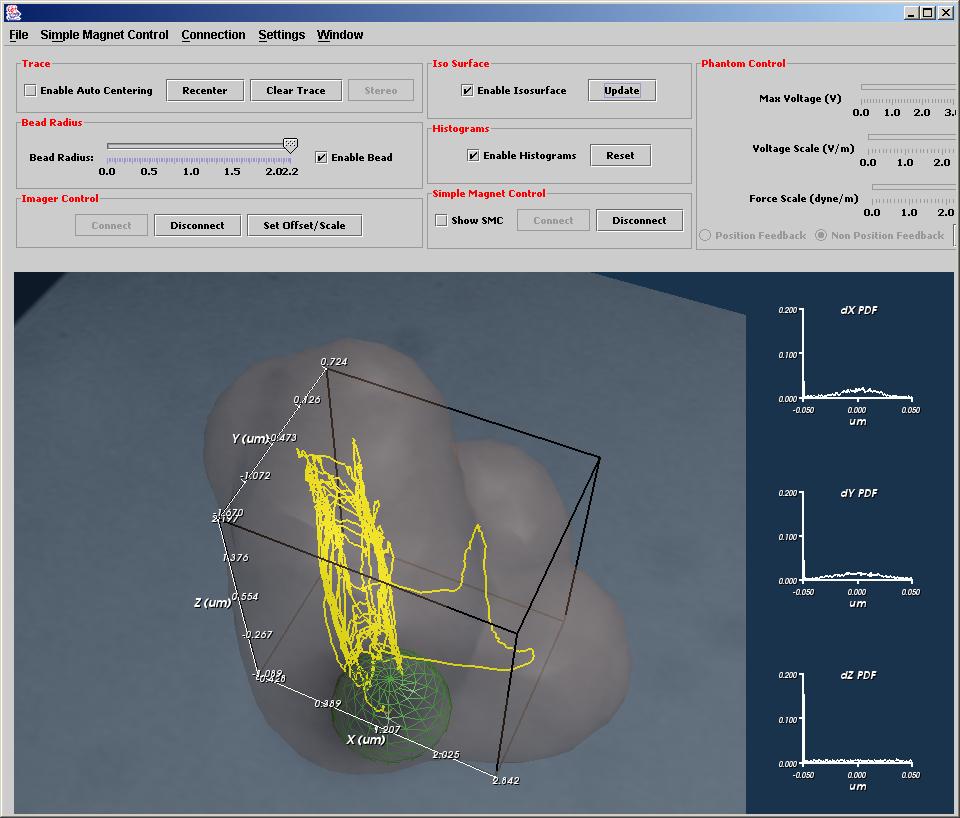
3DFM Concept Video
A fanciful view inside a cell intended to show the concept of the 3DFM. It starts with a view of several microtubules and follows the motion of a bead along one of them. As it zooms in, we can see a representation of a kinesin walking its way along the microtubule, carrying the bead. Using the 3DFM, we hope to be able to track, stall, and measure the forces on kinesin and other motor proteins as they perform their duties within living cells.
The Ciliary Beat
Very high magnification view of cilia beating in real time. Video images were improved with noise reduction and contrast enhancemnent to make this motion visible. 400x magnification was used to relay the image to a newvicon tube camera, and differnetial interference contrast light microscopy to form the primary image. These were human airway epithelial cell cultures obtained from the UNC Cystic Fibrosis Center and grown in our laboratory. Each cilium is about 7 um long, and they can beat at up to 30 times per second, although those shown were much slower.
Cilia Hurricane
Very low magnification video of cultured ciliated airway cells grown in the CISMM cell culture facility. The cilia begin to beat in a coordinated manner that has the effect of moving the media, particles and mucous secreted by the cells in a circle around the culture well. These were first demonstrated by Hirotoshi Matsui and colleagues at the UNC Cystic Fibrosis Center. We use these “hurricanes” to find cultures with good ciliary function.
Big Bead Catch
A 2.8-micron diameter bead attached to slowly-beating cilia is tracked by the laser tracking and position feedback systems of the 3DFM. For the first two cycles, the bead is not tracked; it and the other two beads near the bottom of the image move as the cilia beat. As the tracking feedback kicks in, the background moves while the bead stays fixed in the center of the “+” sign. Note that the point on the bead that is tracked is slightly out of the focal plane of the optical microscope, so the three beads get slightly fuzzy while tracking is operating.
3DFM Prototype Haptic Bead Control
Jeremy Cummings using a force-feedback device to control the magnetic drive on the first prototype of the 3D Force Microscope. This drives a magnetic bead in a square.