Project 3: Force Manipulation Systems
We have developed a new imaging system that enables real-time X-Z imaging simultaneous with traditional X-Y imaging. This is done by arranging a 90º reflecting optic above the specimen within the field of view of the standard epi-fluorescence imaging system. We call this method PRISM, for Pathway Rotated Imaging for Sideways Microscopy. We use a freely positioned reflecting optic that can observe a wide variety of specimens, with standard imaging microscopes, integrating it with light sheet microscopy and atomic force microscopy.
Structural strain information imposed within the cell, cytoskeletal adaptation, and biochemical distributions are all critical for understanding cell response quantitatively. However, structural information during applied stress is limited by our ability to image cells under load. Traditionally, this is done using epi-fluorescence imaging, which provides a “plan-view” image of the specimen. This is limiting because the force (and hence dominant strains) are often in the z-direction. Beyond structural information of mechanical strain, remarkable insight into dynamics of diffusion, transport and binding have been obtained by image correlation spectroscopy (ICS). With CISMM’s recent work on cell force response, and our measurement of nuclear force response, we are positioned to explore the full problem of force transmission from specific ligands to the cytoskeleton to the nucleus, and to study the consequences for remodeling of the cytoskeleton and for gene expression in the nucleus. This requires us to implement high spatial resolution, millisecond time scale imaging in the XZ plane, integrated with our force measurement techniques AFM and 3DFM magnetics.
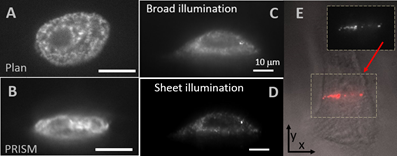
A) Plan view (XY) of mesenchymal stem cell nucleus (Guilak) labeled with GFP histones, with B) widefield PRISM (XZ) view. Scale bars 10 microns. The detail of histones is evident. We expect this to improve further with new lasers specified that allow light sheet imaging for eGFP
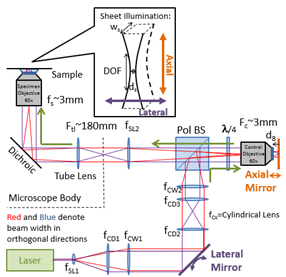
Vertical Light Sheet (VLS) illumination improves imaging of 20nm bead labeled ovarian cancer cells with C) broad illumination and D) vertical sheet illumination. E) Plan view of 20nm bead-labeled cells with brightfield image overlaid with fluorescent sheet image (plan view) showing narrow stripe illumination. F) VLS PRISM view/ 1ms exposure. Right is schematic of VLS optical layout with the lateral mirror providing lateral displacement of the laser illumination, and the axial mirror providing the axial displacement of the beam waist. These two optics, as well as the lasers, will be computer controlled in the new setup to provide automated switching of the observation plane.
This movie shows a side-view of a SKOV, an ovarian cancer cell line, nucleus labeled with a red fluorescent nucleic acid stain as captured with Vertical Light Sheet Sideways Microscopy (VLSSM). Over the course of the movie an Atomic Force Microscope (AFM) cantilever with 5μm bead tip deforms the nucleus with 50nN of force. The movie was recorded at 40fps with simultaneous force data acquired by an AFM.
This movie shows a side-view of the nucleus of a SKOV cell, an ovarian cancer cell line, with the DNA labeled with a red fluorescent nucleic acid stain and and membrane stain in same channel, as captured with Vertical Light Sheet Sideways Microscopy (VLSSM). Over the course of the movie an Atomic Force Microscope (AFM) cantilever with 5μm bead tip deforms the nucleus with 10nN of force. The movie was recorded at 50fps with simultaneous force data acquired by an AFM.