ASAP
A quantitative understanding of the rheological properties of biofluids is of critical imporatance in biomedical contexts ranging from respiratory illness, to heart disease, to fertility. However, typical rheometers require large specimen volumes, extensive pre-analytical processing, and highly-trained operators. We have developed a novel technique for measuring the time dependent elastic properties of microliter quantities of material. We are developing this system as a blood clot diagnostic and have begun studies exploring its use in mucus rheology studies. This system magnetically actuates arrays of magnetic posts that mimic biological cilia. We call this system Actuated Surface Attached Posts (ASAP). The goal is to take a magnetic force assay from the microscope into a disposable strip for clinical diagnosing clot disorders and characterizing mucus.
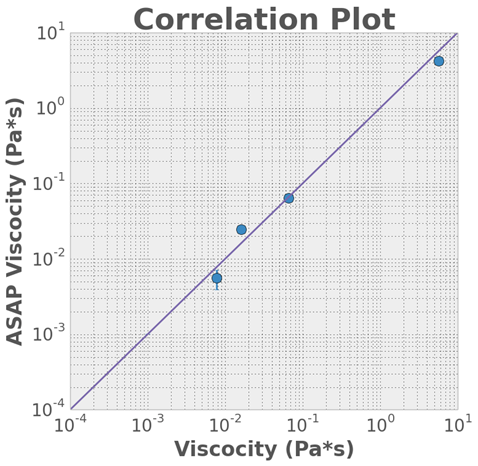
(Left) ASAP posts. We have demonstrated consistent post fabrication using track-etched polycarbonate membranes as a template for PDMS molding and nickel electrochemical deposition to form our magnetic posts. The lighter grey tops of the posts are nickel shells surrounding a flexible PDSM rod. The ASAP posts are actuated with electromagnets. (Right) Correlation plot of ASAP vs. conventional rheometer.We have demonstrated that ASAP can act as a working rheometer for over 3 orders of magnitude in viscosity
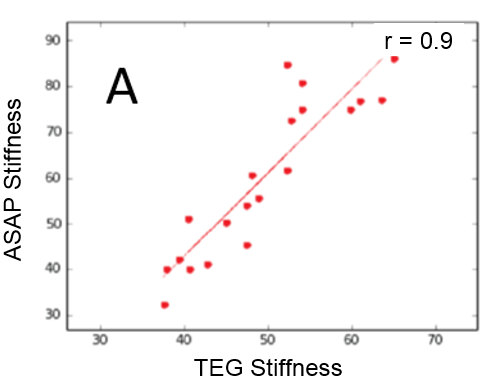
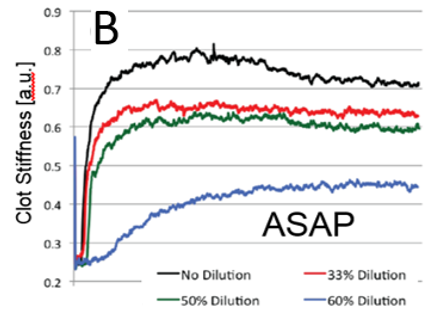
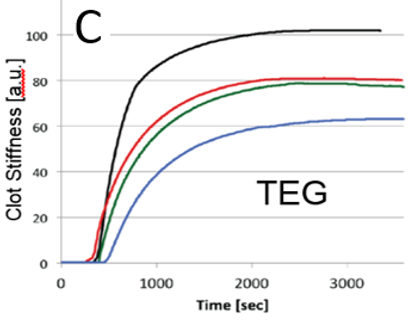
(A) Correlation plot between the ASAP and TEG systems. ASAP data (B) and TEG data (C) for a parallel whole blood dilution as-say. Note: TEG software applies a smoothing process to the raw data.
The three applications of our ASAP technology are in clot diagnostics, studies focusing on shear activation of platelets, and in patient mucus characterization. For clot diagnostics, our collaborator Prof. Herb Whinna, head of the McLennan Clinical Diagnostic Laboratory at UNC Hospitals, is partnering with us to develop the diagnostic assay of clot pathology for surgical patients. This technology also has applications for small animal studies–blood specimens are small and valuable. This project teams with a start-up company Rheomics, Inc. that has licensed the technology for commercialization. We are also developing an ASAP based platelet shear-activation assay. We are partnering with Prof. Wolfgang Bergmeier in investigating anti-platelet strategies focusing on inhibitors to αIIbβ3. We have a longstanding interest in mucus rheology through the Virtual Lung Project, and more recently in our work with Katharina Ribbeck’s lab focusing on cervical mucus. The aim here will be in developing ASAP as a tool for testing cervical mucus rheological properties as a biomarker predicting risk for preterm birth.